
- 285
- 113 565 845
NurdRage
Canada
Registrace 24. 11. 2008
A channel run by science nerds for science nerds hailing from the country of Canada.
We demonstrate science experiments for all levels, from kitchen chemistry to advanced synthesis.
Because what we do is very expensive due to the cost of chemicals and lab equipment, we would appreciate your support at our pateon account: www.patreon.com/NurdRage?ty=h
Bitcoin donations can be made to: 1NurdRAge7PNR4ULrbrpcYvc9RC4LDp9pS
For our viewers in china, you can also find our equivalent channels at
www.weibo.com/nurdrage
space.bilibili.com/286479171/#/
We demonstrate science experiments for all levels, from kitchen chemistry to advanced synthesis.
Because what we do is very expensive due to the cost of chemicals and lab equipment, we would appreciate your support at our pateon account: www.patreon.com/NurdRage?ty=h
Bitcoin donations can be made to: 1NurdRAge7PNR4ULrbrpcYvc9RC4LDp9pS
For our viewers in china, you can also find our equivalent channels at
www.weibo.com/nurdrage
space.bilibili.com/286479171/#/
Lab notes - Making Oleum - 20% yield improvement - (May 4th - 2024)
More lab notes on making sulfur trioxide and oleum. This time i was able to get up to 20%-25% yield with improved heating with better insulation. Also found out that precise temperature control is important and that the polymerization of the sulfur trioxide phases can be easily initiated by smearing it with a pipette on air.
Previous videos:
Purification of Sulfuric Acid by Distillation Revisited: czcams.com/video/0Gb9rM9BJ8I/video.html
Lab notes - Making Oleum - Success (part 1): czcams.com/video/wB2zzm8VP9Y/video.html
Lab notes - Making Oleum - 20% yield improvement: This Video
Donate to NurdRage!
Through Patreon (preferred): www.patreon.com/NurdRage
Through CZcams Memberships: czcams.com/channels/IgKGGJkt1MrNmhq3vRibYA.htmljoin
Twitter: NurdRage
Reddit: www.reddit.com/r/NurdRage/
Instagram: nurdrageyoutube
Previous videos:
Purification of Sulfuric Acid by Distillation Revisited: czcams.com/video/0Gb9rM9BJ8I/video.html
Lab notes - Making Oleum - Success (part 1): czcams.com/video/wB2zzm8VP9Y/video.html
Lab notes - Making Oleum - 20% yield improvement: This Video
Donate to NurdRage!
Through Patreon (preferred): www.patreon.com/NurdRage
Through CZcams Memberships: czcams.com/channels/IgKGGJkt1MrNmhq3vRibYA.htmljoin
Twitter: NurdRage
Reddit: www.reddit.com/r/NurdRage/
Instagram: nurdrageyoutube
zhlédnutí: 16 807
Video
Lab Notes - Making Oleum - Successful - (April 21-2024)
zhlédnutí 33KPřed 14 dny
These are my lab notes trying to make Oleum or Fuming Sulfuric Acid by amateur means. Turns out it is possible to do it with just a ceramic hotplate, sulfuric acid, and sodium bisulfate. Along with standard laboratory glassware. But so far the yield is around 13%. I'm trying to optimize it. The procedure is simply heat the sodium bisulfate on the ceramic hotplate and pyrolyze it until it become...
Purification of Sulfuric Acid by Distillation - Revisited
zhlédnutí 46KPřed měsícem
In this video we purify sulfuric acid again using distillation but improve our technique using a ceramic hotplate stirrer and PTFE tape to seal the joints. I start by highlighting the inherent dangers associated with handling sulfuric acid and stress the importance of safety precautions, including wearing protective clothing and performing the procedure in a well-ventilated area or fume hood. I...
Do your own Gamma Spectroscopy with the Radiacode 103
zhlédnutí 24KPřed měsícem
Buy the RadiaCode here: 102.radiacode.com/NurdRage In this video we review the radiacode 103, a handheld gamma ray spectrometer. We also as explore basic aspects of gamma ray spectroscopy. Geiger counters measure radiation, and they're easy to buy online. But all they do is measure how much radiation there is. The Radiacode is a gamma spectrometer and can measure the energy of each individual g...
Thanks for 2023, Channel Updates, Plans, and Thoughts
zhlédnutí 19KPřed 4 měsíci
Thanks for watching! Donate to NurdRage! Through Patreon (preferred): www.patreon.com/NurdRage Through CZcams Memberships: czcams.com/channels/IgKGGJkt1MrNmhq3vRibYA.htmljoin Other CZcams channels that i think are amazing: @That_Chemist @theCodyReeder @ExplosionsAndFire @nomcopterlabs @ChemicalForce @AppliedScience @Chemiolis @chemdelic @LabCoatz_Science @AdvancedTinkering @THYZOID @NileRed Twi...
Promethean Matches - The Ancestor to Modern Matches
zhlédnutí 66KPřed 4 měsíci
In this video we make the ancestor to modern matches before phosphorus: The Promethean Match The operating principle is that sulfuric acid would react with potassium chlorate and sugar to catch fire. So to turn it into a match, the sulfuric acid was stored in a glass capsule that was broken and mixed with potassium chlorate and sugar. This would ignite and set fire to the paper roll it was pack...
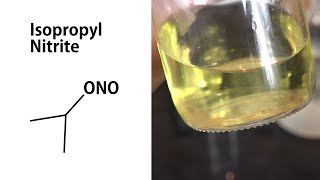
Make Isopropyl Nitrite without Sodium Nitrite
zhlédnutí 23KPřed 4 měsíci
In this video, we make isopropyl nitrite without using sodium nitrite. Typically, amateur chemists use sodium nitrite, hydrochloric acid, and isopropyl alcohol for this process, but sodium nitrite is becoming harder to obtain. This sodium nitrite-free pathway goes through nitrosylsulfuric acid which can be made using more easily obtained fuming nitric acid and sulfur dioxide. To make fuming nit...
This Candle MAKES Oxygen and Started a Fire on a Space Station
zhlédnutí 153KPřed 4 měsíci
In this video we make an Oxygen Candle that actually MAKES oxygen as it burns. It's also called a chemical oxygen generator. To make it, 60g of potassium chlorate are mixed with 30g of iron metal powder and 2g of calcium oxide. The powders are thoroughly mixed and initiated by direct flames or a sparkler. The reaction burns the iron powder and generates enough heat to decompose the remaining po...
How to Make Your Own Flameless Ration Heaters and the science of how they work.
zhlédnutí 54KPřed 5 měsíci
In this video we show how Flameless Ration Heaters and self heating food containers work. Inside of them is a water activated exothermic chemical reaction. The most common one is magnesium iron alloy and salt. When water is activated it produces magnesium hydroxide and plenty of heat. Less common mixtures include sodium hydroxide and aluminum, magnesium metal and sodium bisulfate, aluminum and ...
Lab Notes - Failure in Making Sodium Nitrite
zhlédnutí 20KPřed 6 měsíci
In this video i synthesize nitrosylsulfuric acid and isopropyl nitrite, and i tried to make sodium nitrite but i failed. My original goal was to make sodium nitrite. Sodium nitrite sets itself apart from the more common sodium nitrate by having one less oxygen atom, which makes it quite unstable under various conditions. This particular challenge intrigued me, and I thought I'd give it a try, e...
Make Potassium Permanganate
zhlédnutí 45KPřed 7 měsíci
In this video, we explore the preparation of potassium permanganate, a valuable oxidant with a striking purple color. Starting with 43g of manganese dioxide, often obtained from greensand filter media, and 25g of potassium chlorate, the chemicals are thoroughly mixed. A can made of iron is chosen as the reaction vessel due to its resistance to the highly corrosive mixture. All reagents are mixe...
Chemical Thunderstorm in a Beaker
zhlédnutí 24KPřed 7 měsíci
In this video we make a chemical thunderstorm in a beaker using manganese heptoxide. I wanted recreate the test tube thunderstorm on a larger scale, adding sound for a more immersive experience. The setup involved using a glass funnel to carefully pour sulfuric acid into ethanol and form a layer of it in the bottom. Sprinkling potassium permanganate into the mix led to the formation of manganes...
Lab Equipment Repair: Broken Stirrer Encoder Disc in Hotplate Stirrer
zhlédnutí 14KPřed 7 měsíci
Lab Equipment Repair: Broken Stirrer Encoder Disc in Hotplate Stirrer
Lab notes - Making Potassium Permanganate
zhlédnutí 77KPřed 7 měsíci
Lab notes - Making Potassium Permanganate
Find the Concentration of Hydrogen Peroxide by Titration with Potassium Permanganate
zhlédnutí 41KPřed 10 měsíci
Find the Concentration of Hydrogen Peroxide by Titration with Potassium Permanganate
Make Sodium Oxalate - Primary Standard for Analytical Chemistry
zhlédnutí 24KPřed 11 měsíci
Make Sodium Oxalate - Primary Standard for Analytical Chemistry
Lab Equipment: Rotary Evaporator or "Rotovap"
zhlédnutí 50KPřed rokem
Lab Equipment: Rotary Evaporator or "Rotovap"
How Does Aqua Regia Dissolve Gold and Platinum?
zhlédnutí 80KPřed rokem
How Does Aqua Regia Dissolve Gold and Platinum?
Make Sodium Hydroxide by Electrolysis with a Nafion Membrane Cell
zhlédnutí 45KPřed rokem
Make Sodium Hydroxide by Electrolysis with a Nafion Membrane Cell
Make a Nafion Divided Membrane Cell for Electrolysis
zhlédnutí 52KPřed rokem
Make a Nafion Divided Membrane Cell for Electrolysis
Lab Notes: Making Copper Sulfate and Electrobonding Wires
zhlédnutí 100KPřed 2 lety
Lab Notes: Making Copper Sulfate and Electrobonding Wires
Efficiently Recover Nitric Acid and Copper Metal From Copper Nitrate Wastes
zhlédnutí 133KPřed 3 lety
Efficiently Recover Nitric Acid and Copper Metal From Copper Nitrate Wastes
Nitric Acid Concentration and Purification (Azeotropic and Fuming)
zhlédnutí 135KPřed 3 lety
Nitric Acid Concentration and Purification (Azeotropic and Fuming)
Make Nitric Acid by Thermal Decomposition of Copper Nitrate
zhlédnutí 70KPřed 3 lety
Make Nitric Acid by Thermal Decomposition of Copper Nitrate
Lab Notes - August 31st - Nitric Acid from Calcium Nitrate and My Hotplate Failed, Again.
zhlédnutí 41KPřed 3 lety
Lab Notes - August 31st - Nitric Acid from Calcium Nitrate and My Hotplate Failed, Again.
Make Nitric Acid from Sodium Bisulfate and Sodium Nitrate
zhlédnutí 132KPřed 3 lety
Make Nitric Acid from Sodium Bisulfate and Sodium Nitrate








Could u use calcium hypochlorite from say pool shock and potassium chloride in a cell like this ?
hi Could you make potassium perchlorate?
This little video of predictive programming brought to you by The World Economici Forum, Pfizer, and the ADL.
A wayward thought... aerosolized hydrogen peroxide is suspected to cause SO3 formation in the environment. I'm not sure what method would be best for aerosolizing H2O2, but there are peroxide fogging machines for sale (not sure how they're different from water foggers); I wonder whether one of those cheap atomization discs might work. Also, the logistics of combining gases might be an issue. In my experience, the vapor from those atomizers tend to be cold and fall, so perhaps just introducing concentrated peroxide vapor from somewhere above the distilling flask would help, especially at the later stage, when more water begins to form in the distilling flask, anyway.
A second (possibly better) wayward thought... perhaps a dessicant (another sulfate?) could be added, if moisture is the reason the reaction is stopping short.
A third wayward thought... perhaps you could use a two necked receiving flask and a glass rod, or maybe dangle (with string made of teflon tape) a seeding spot, near the end of the condenser, as an indicator, so that you aren't forming crystals that block the condenser.
No! Not getting a job!! 😂😂😂
If they could get 1 amp, from each battery at 4 volts this type of cell could be combined to make a 48v battery with 12 cells pushing out 12 amps x this by 100 and you have a very impresive power source. More than enough to charge a larger battery for however long each cell lasts, put everything in a clear packaging allowing it to absorb natural light and you have a solar panel that generates power all night and more during the day. It is just getting it up to 1 amp which means experimenting with solar panel used and size.
My favorite part of the video was the sneaky diss on Nigel at the end. 😂
Yo!!
What about using a top-loading kiln with boiling chips? Instead of closing the lid of the kiln, buy some rockwool or ceramic-fiber insulation so you can have the glass sticking out of the top. If stirring is a must, then try wrapping the reaction flask and the top edge of the hot plate in ceramic wool. If that doesn't work, I'd try building a box out of hardware cloth and coating it in refractory cement. If you make it so it can drop down over the flask's neck and support it on some plain old bricks or those screw lifts, you could even add some supplemental heat from a butane torch firing up from underneath.
always love your work!
Wait. You exist?!
keep talking nerdy to us
So glad youre back! 🤩
Oleum? Darn near killed um!
Could this method be adapted to obtain metallic potassium? thanks 🥰
I initially misread the title and thought the video was about the zero-calorie fat substitute, turns out that's Olestra Definitely don't confuse the two
А нельзя ли приблизительно рассчитать теоретически, в каком интервале будет происходить реакция с образованием SO3, а в каком другая реакция? Через энергию Гиббса, например
Awesome
I didn’t realize you were back making videos. I missed a bunch I’ll have to go back over. You made the best early CZcams science content
can you use an open flame for heating? reason: cheaper and more common & the solution doesn't require stirring. problem: temperature control.
Could roughing up the glass at the end of the condenser provide nucleation sites for the crystals to form again after melting?
which scintillators have best value?
Thanks for this...I loved Chemistry at school, especially the lab work and watching demonstrations. It's like being back there, accompanied by the same sense of wonder
i was going to suggest Nichrome wire raped on the spigot but google says it may corrode in the presence of air or if heated this is a contaminated idea for the algorithm...
I've been following for many years and do my best to like every video I see. I'm sadly too poor to be able to give to a Patreon,
I would like to see you work on your hot plate. That kind of thing is interesting to me.
awesome vid
it may be viable to use a cooking hot plate as they get red hot on full power, theyre dime a dozen and precision is not necessary here so a simple thermocouple under the flask should be sufficient for control, good research as always
This reminds me of the making sodium video. Using oleum to pre dry the sulfuric acid seems like a logical step. I also think that a larger ratio of bisulfate should be used since the catalyst has a maximum water capacity.
It looks like you're losing some sulfur trioxide out of the vacuum adapter. Have you consider passing this through water to capture it as sulfuric acid or is not worth it?
hey youtube: he exists
Is there a non reactive crystal that you could "glue" to the end of the exit nozzle that would help create a seed site for the polymerization to occur? Or does it have to occur naturally?
You exist. Congratulations!
I always fancied a stainless steel apparatus fed with sulphur powder and air and producing SO3 over a V2O5 catalyst fused onto a mesh. Dissolving the product SO3 in strong sulphuric acid would have made a stronger acid then an oleum/acid mix. Realistically my need for oleum didn't justify the cost :(
Don't worry, you exist!!!
... throw the solid SO3 into some paper please o.o
What about a carbothermal reduction of the gallium oxide?
How about separating lead and antimony from ammunition to get pure lead?
Wicked looking waxy solid.
Yeaaaah now we we can make sulfuric acid
Great improvements! I think some way of infrared heating may work directly through glass and heat up crystallized oleum directly. Would be faster than heating the flask.
Three comments: 1) Have you tried improving yield by adding aluminium (Al3+) ions to the reaction? Heating potassium-aluminum-sulfate used to be the alchemists' way of making sulfuric acid. I don't know, if the products sold as aluminum sulfate Al2(SO4)3 are indeed anhydrous, but they should give up their water readily at boiling sulfuric acid temperature. Anyway, I think Al3+ ions might also act catalytical and a small percentage of added aluminium sulfate might lower the melting point of your sodium bisulfate (and having a small amount of potassium bisulfate in the mix might lower the melting point even more). 2) With a substances as hygroscopic as oleum/sulfur trioxide I'd probably prefer to exclude moisture from the distillation apparatus using a moisture trap - but I can understand if for concerns of pressure build up you do without a trap. 3) Heating device: Here really a metal bath made out of a low melting alloy might make sense. You could use a normal electric hotplate as heat source. Heat transfer will be much better if you can (partially) immerse the reation flask into the bath rather than having it stand on top of a plate.
I wonder if a stamped metal (or other material) shape might help to collect the crystals? something that folded and gripped the pipette feeding the collection vessel, that served as either a heat sink (via convection, may cool the solution that drips off) or a collection point itself.
Dear NSA, I’m not a terrorist, just a aspiring chemist
If I'm understanding this reaction correctly, initial chemicals could be dissolved in separate containers and stored for later use so long as the sugar/dextrose is not added to the mixture until it's time to do the final mirroring reaction? And the silver nitrate solution should be maintained in dark glass. And potassium hydroxide should be a suitable substitute in the event sodium hydroxide is not available?
Similar to the sodium vids, would an oleum starter work for better yield? IE get as close to 100% H2SO4 (and a bit over) to ensure there is no water to ruin yield? You could then keep recycling that for your pyrolysis?
future nurd rage sent me
free comment for all the years of content
Commenting for proof of existing
Nice work ! :D